You flip the switch and the light goes on. You find yourself wondering how electricity really works. What is electricity? Where does it come from? And how is electricity stored in batteries? These are only a few questions you might be wondering about. If you’ve always been burning to learn more about how electricity really works, you’re in the right place. So, how does electricity work? Here’s a brief overview.
A Brief History of Electricity
People have been interested in using electricity for thousands of years. The first records of electric use come from the Roman Empire.
Back in the ancient times, Pliny the Elder (an author and natural philosopher) and Scribonius Largus (a court physician) spoke of how patients were advised to touch electric fish such as electric catfish or rays to cure ailments like gout or headache. The hope was that the electric shock would heal these conditions.
It was only in the 18th century things started to pick up. Particularly, when Benjamin Franklin flew a kite in a lightning storm. By doing this, Franklin proved that lightning was a form of electricity – static electricity, to be exact. Although others are also credited with the discovery of electricity, Franklin’s story remains the most infamous.
Alessandro Volta made the first battery that could continually sustain electric flow in 1800. It was called the voltaic pile. Soon after, Michael Faraday invented the electric motor in 1821.
The 1900’s started saw more inventions come to life. From Thomas Edison with the incandescent lightbulb and his managerial counterpart, Samuel Insull, who made electricity affordable for more people.
After this, Alexander Graham Bell is known for inventing and patenting the first telephone. And lastly, there’s Nikola Tesla, who’s is well-known for his studies on alternating current.
Why Are Some Materials Conductive?
To understand this topic, it’s good to know about basic atomic structure. You might remember this information from school.
An atom’s nucleus (also known as the center or core), is made up of protons and neutrons. Protons have a positive charge and neutrons don’t have any charge. On the outside of the nucleus, there are electrons, which have a negative charge.
The protons in the nucleus help to keep the electrons within the atom by exerting positive forces. Electrons, being negative, are attracted to the positive forces of the protons, seeing as opposites attract. Because of this, electrons are always found in the space outside an atom’s nucleus.
While some electrons can be found close to the core of the atom, many are further away. Bohr’s atomic model illustrates this concept in a simple way. The model shows a number of concentric circles similar to a dart board or shooting target. The “bullseye” in the model represents the atom’s core, as it’s found right in the center. The layers of concentric circles are called “atomic shells” and electrons are illustrated as dots on the lines of the circles to show in which shell they are located.
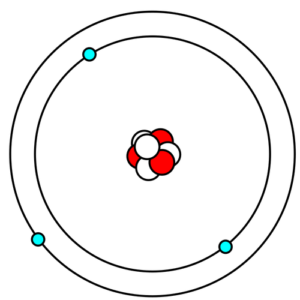
This is an example of Bohr’s atomic model with the electrons orbiting the nucleus in different atomic shells.
Once you have a basic understanding of atomic structure, it will be easier to know conductors and insulators work.
Insulators
Insulators can’t conduct electricity. When looking at the atoms of insulators, you will note that the electrons are kept close to the nucleus.
With all the electrons so close to the nucleus, there aren’t any electrons that can venture into shells that are far enough from the center to help insulators conducts electricity. Electrons can be shared by atoms, but not when they are too close to the nucleus.
Conductors
Conductors are materials that can conduct or “transport” electricity. The atoms of conductors have free electrons. Free electrons can escape the orbit of their neutron if you add energy. This sharing of electrons is what causes a material to be conductive. Electrons push each other away (or repel one another due to shared negativity). Because the free electrons can escape the orbit, the electrons start flowing, helping electric energy to be conducted.
The fact that electricity is conducted on the atomic scale by electrons is interesting, as this explains why the words “electrons” and “electricity” are related. Thus, electric energy can simply be explained as energy from electrons flowing through wires.
How Is Electricity Generated?
This is just the general concept of how electricity is generated. The process works almost the same for all the thermal power plants.
- Fuel is burned in a furnace, which heats a boiler filled with water. Another way to create heat is by splitting atoms. This is called a nuclear power plant. The most common fuels used in power plants include coal, natural gas and petroleum.
- Heat from the burning fuel causes the boiler water to boil. The boiling water turns to steam.
- The steam then moves to a turbine, which looks like a fan with multiple blades. As steam moves through the turbine, it starts to turn.
- The turbine is connected to an axle. On the other end of the axle is a generator. The generator is responsible for creating electricity. It turns the energy from turning, known as kinetic energy, to electric energy.
You might remember from school that energy can’t be created or destroyed, only converted from one from to another. Because of this, the process of generating electricity isn’t creating energy. Instead, kinetic energy, or “movement energy” is simply converted to electric energy.
There are other ways to generate electricity. Examples include solar, hydroelectric and wind powered electricity. Both hydroelectric and wind power work much like thermal power plants. The only difference is that wind or water is used to turn turbines rather than steam.
Solar power plants get their electricity from the sun with the help of photovoltaics. This process of producing solar power is very different from other methods of generating electricity and won’t be discussed in this article.
How Does Electricity Get to My House?
After electricity has been generated, it moves though wires. First it goes to a step-up transformer. When electricity moves through wires, it loses energy. To solve this problem, the flow of electricity is sped up. Higher-Voltage results in less loss of electric energy while moving through wiring.
Next, electricity moves to pylons. Pylons are big metal towers. They carry the high-voltage electricity to step-down transformers. Seeing as high-voltage electricity isn’t suitable to use directly in your house, a step-down transformer makes electricity low-voltage again. Finally, after going through the step-down transformer, the electricity can be used in your house or business.
How Do Batteries Work?
Batteries are made up of electrochemical cells. Either one or multiple cells can be used for a battery. The term “cell” doesn’t refer to a microscopic organism in this case. Instead, electrochemical cells are devices that generate electricity from a chemical reaction inside the cell.
Electrochemical cells have two electrodes (positive and negative), separated by an electrolyte – also called an ionic liquid.
The negative end of the battery is called the anode. Chemical reactions cause a buildup of electrons at the anode. As you know, same charges push each other away, so this buildup of electrons causes charge.
The charge travels from the negative terminal of the battery to the positive terminal. The positive terminal is called the cathode. Here, the chemical reaction receives electrons, after they have gone through the circuit.
The battery is filled with electrolyte (also called the ionic liquid). This liquid ensures the electrons don’t simply travel through the battery to the cathode, allowing the charge to flow through the wiring instead.
When you have a rechargeable battery – or secondary cell – this reaction happens in reverse whenever you charge the battery. In other words, all the electrons that flowed to the cathode moves back to the anode. Reversing the process allows you to use the battery’s charge until you have to reverse the process again through charging.
What Is the Difference Between Amps, Volts and Watts?
There are different ways to measure electricity and many find it confusing. Here are some electrical units and what they measure.
Amps
Amps is the flow of electricity, or as you now know, the number of electrons that pass a given point in one second.
Volts
Volts is the amount of force that the electricity is flowing with.
Watts
Watts is the power with which the electricity is flowing. Amps multiplied by Volts equals Watts
Conclusion
How electricity works is a long and complex subject, this was simply an overview. Learning how to use electricity has been one of the most fundamental discoveries in science. Almost every other industry, from healthcare and hospitality, to the automotive industry and manufacturing depend on electrical power.
Understanding how electricity works helps everyday individuals know more about the energy that powers our lives.



